
Global Trends in R&D. Overview through 2020
15 June 2021
This report from IQIVA examines the trends in pharmaceutical research and development through the end of 2020. It provides an analysis of initiated clinical trials, including the impact of the pandemic and COVID-19-specific research. Research on clinical development productivity is also presented, as well as profiles of the R&D pipeline, R&D funding trends, and characteristics of new drug approvals and launches.
Report Summary
In 2020, the COVID-19 pandemic upended life globally and yet not only did research and development activities largely continue – albeit with disruption – they also resulted in the development of vaccines that are already reaching more than 600 million people. The key drivers of these unprecedented results are visible in benchmarks and trends of various aspects of R&D, providing insight into what other improvements are already underway or could be possible.
- Funding for early- and late-stage R&D and strategic transactions increased significantly in 2020; aggregate R&D spend for the top 15 companies reached a record high.
- The number of first-time launches of novel active substances (NAS) reached an all-time high of 66 in 2020 – with success for treatments in oncology and rare diseases.
- Clinical trial starts increased 8% in 2020, similar growth to the prior three years, with oncology trial starts reaching historically high levels.
- Overall, clinical trial activity recovered from mid-year 2020 to levels above 2019 – even without COVID-19 trials.
Key Findings
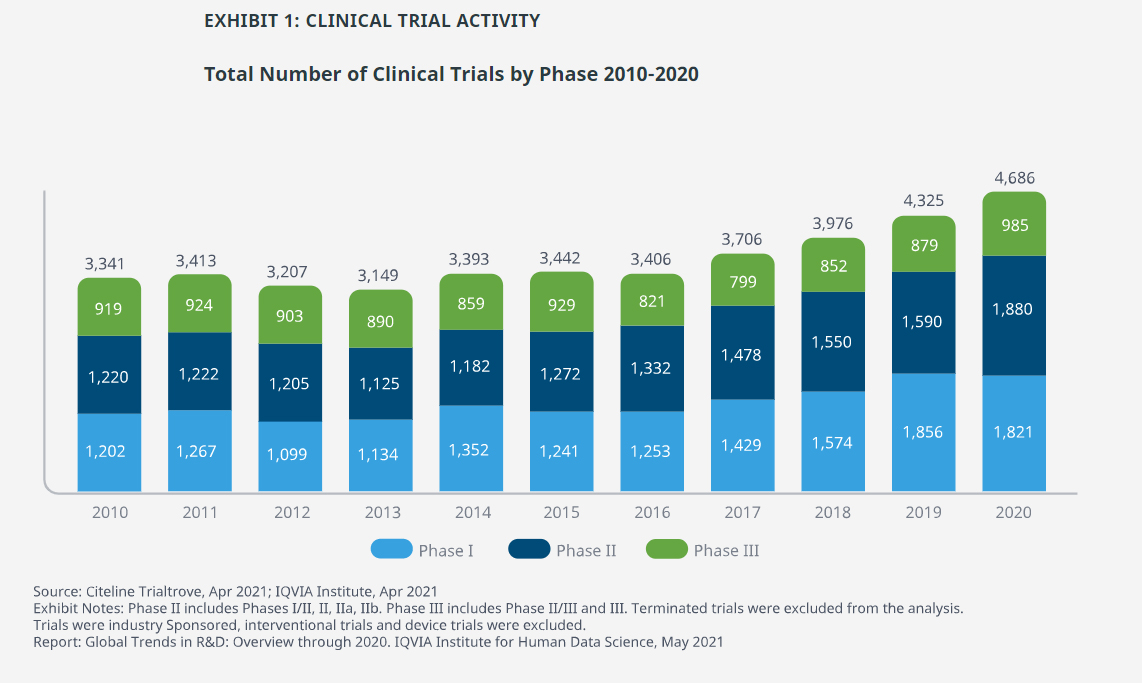
- Despite significant societal disruptions from the COVID-19 pandemic, overall clinical trial activity rose 8% in 2020, the fourth consecutive year with an increase above 7%.
- During the course of 2020, monthly trial starts fell significantly in the first quarter but recovered from mid-year to higher levels than in 2019 even without the COVID-19 trials.
- While the intention to start trials continued with relatively small disruption, the operation of trials — including recruiting patients and completing studies — may have been disrupted differently, and these trends should be interpreted with caution.
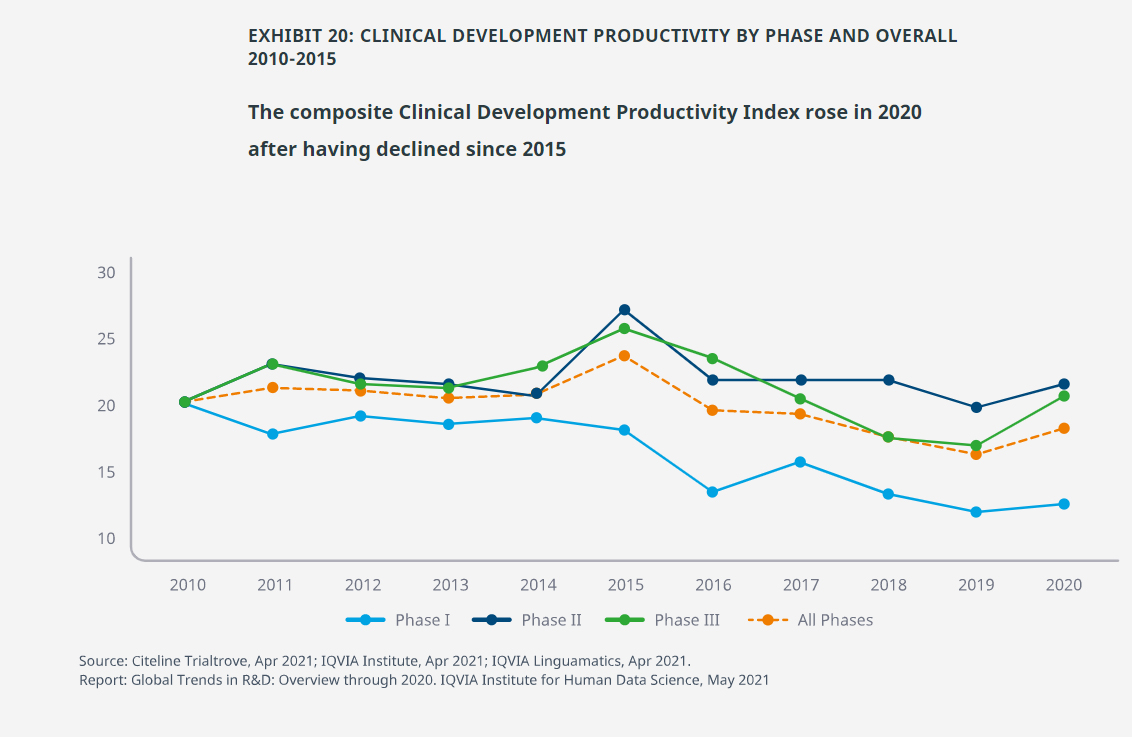
- Most diseases areas have been declining in clinical development productivity – a composite metric of success rates, clinical trial complexity and trial duration – over the past decade, which embeds the challenges of achieving clinical results in well-satisfied traditional disease areas such as cardiovascular and endocrinology, as well as shifts in research priorities to more difficult areas such as oncology and rare diseases.
- The increase in the clinical trial productivity index in 2020 was mostly due to an improvement in Phase III trials, widening the gap with Phase I trials, which score significantly lower with this index.
- Phase II trials have consistently been above the overall index as success rates have been trending up and durations have been trending down, even as complexity has been rising in Phase II as rising numbers of endpoints and eligibility criteria are attributes of these trials.
- Productivity remains below historic levels as success rates are below the long-term average, while complexity attributes of trials are generally rising, as are trial durations in many diseases.
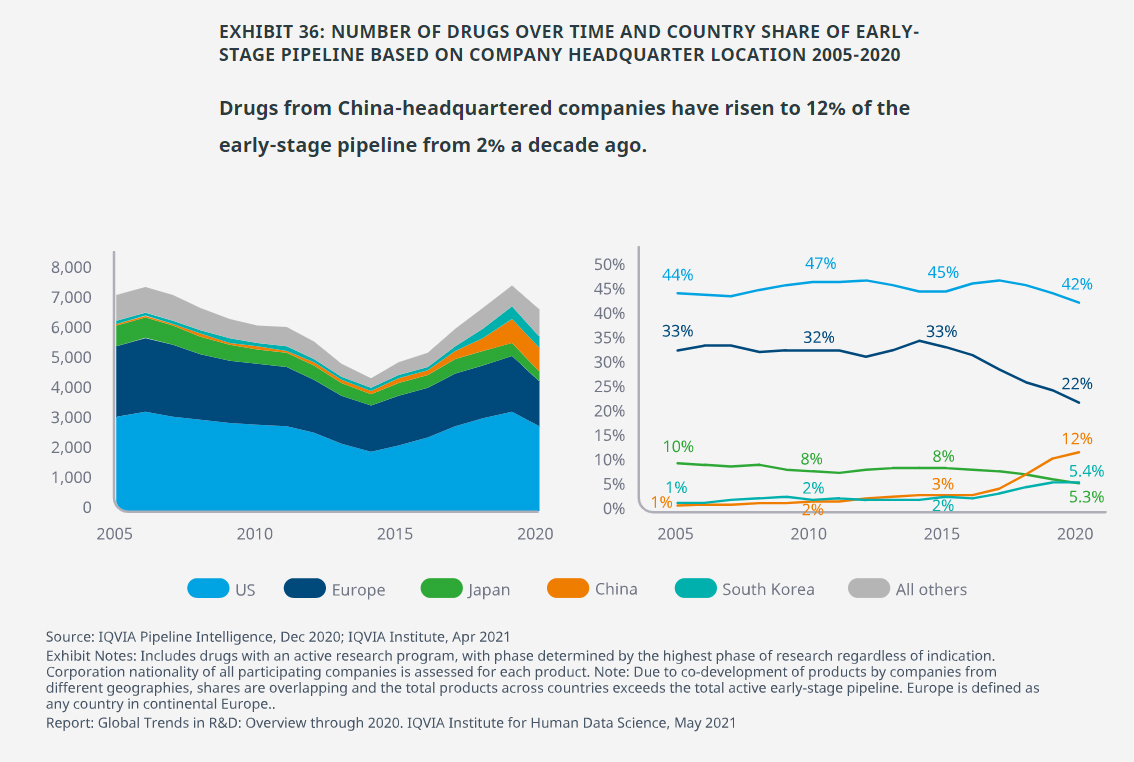
- The U.S. share of global early-stage R&D has remained relatively stable over the past 15 years.
- Europe’s share has declined from 33% to 22% over the past five years, while the absolute number of active programs declined by 175 — from 1,604 to 1,429.
- Products from China-headquartered companies now represent 12% of the early-stage pipeline, up from 2% a decade ago.
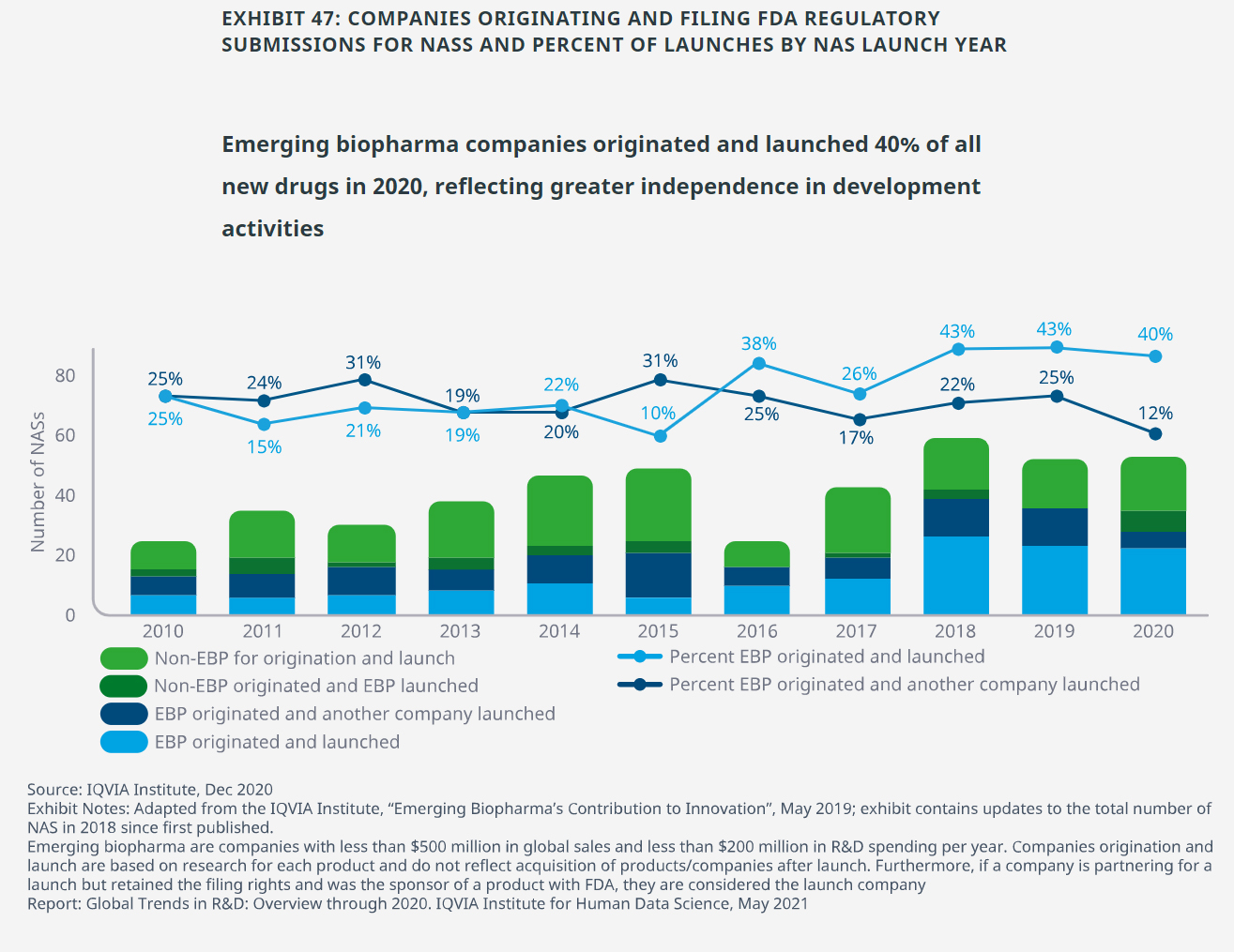
- Traditional development and launch models for innovative medicines relied predominantly on large or mid-sized companies, which often partnered with or acquired smaller companies that originated the medicine.
- That traditional pattern has largely changed in recent years with the last three years having more than 40% of NAS originated and launched by emerging biopharma companies (those with less than $500 million in sales and less than $200 million in R&D spend per year).
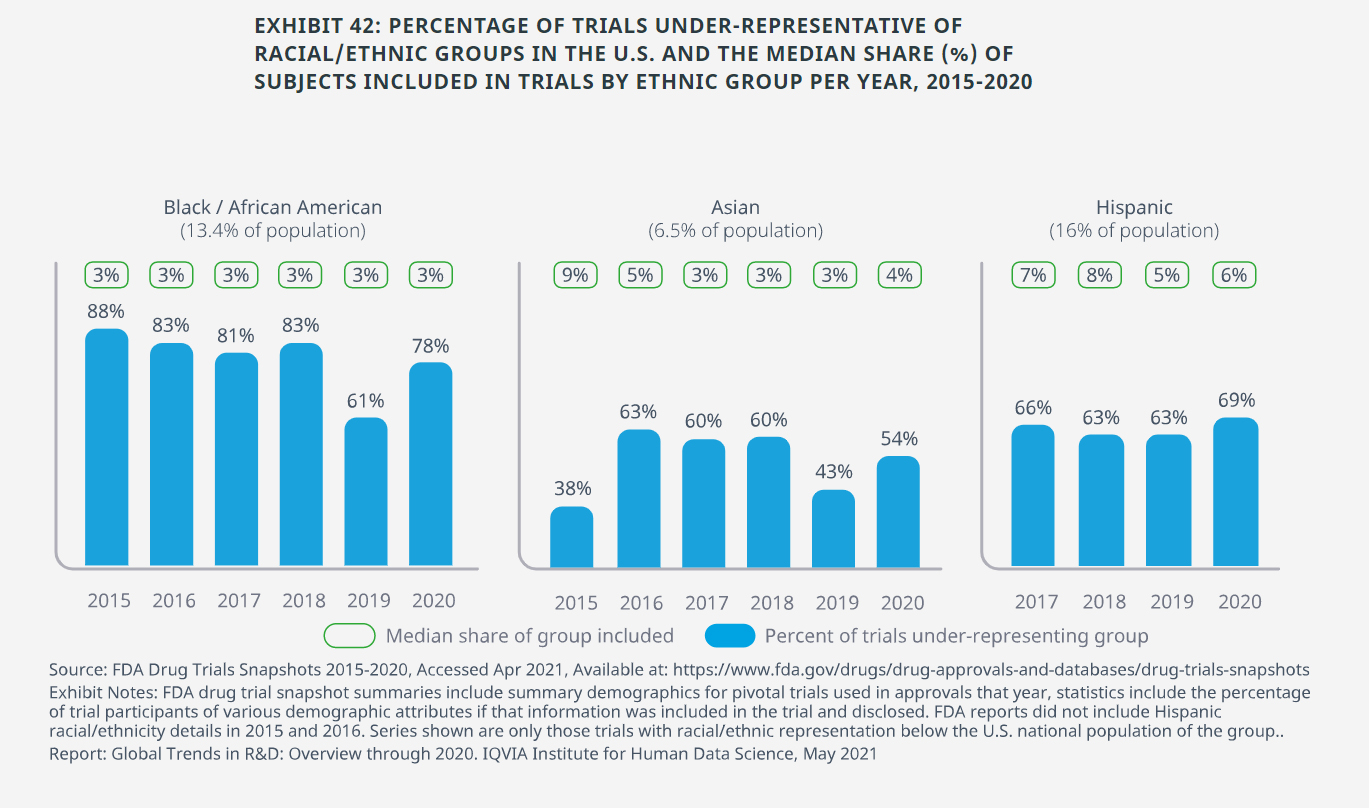
- African Americans or races identified as Black account for 13.4% of the U.S. population, while the clinical trials used to approve new medicines had a median participation of only 3% in the past six years and were under-representative 79% of the time from 2015 to 2020.
- Persons of Asian descent are estimated to comprise 6.5% of the U.S. population, but only in 2015 was the median above this threshold, and 52% of trials in the past six years that were used by the FDA to approve medicines had under-representative participation.
- Hispanic or Latino ethnicity is estimated at 16% of the U.S. population, and consistently the median participation in trials is less than half that level, though patients may not always identify themselves.
- Interpretation of race/ethnicity data of approval trials and the representation of the U.S. population is complicated by the role that ex-U.S. subjects often play in those trials that are executed globally.
Our news
-
14 March 2024
-
26 February 2024
-
NovaMedica team wishes you a Merry Christmas and a Happy New Year!
26 December 2023
Media Center
-
New breast cancer guidelines call for first mammogram at age 40 but stop short of annual screening
08 May 2024
-
WHO proposes recommendations for preventing nitrosamine contamination
07 May 2024
-
Batyr Berdyklychev is now head of the WHO office in Russia
07 May 2024
-
Russia expects more medical tourism due to the world’s first drug for ankylosing spondylitis
07 May 2024